An Avid roadmap for early development—from sequence to a clinic-ready IND package—built around CMC strategy, CLD, and PD decision points.
Moving from a confirmed DNA sequence to an Investigational New Drug (IND) application is a series of deliberate, connected choices—scientific, technical, and operational. The fastest programs aren’t the ones that rush; they’re the ones that align Chemistry, Manufacturing, and Controls (CMC) strategy early, de-risk critical assays, and make data-driven handoffs across development phases. At Avid, we think about “DNA to IND” as a disciplined path with clear milestones, integrated technical teams, and documentation readiness built in from day one.
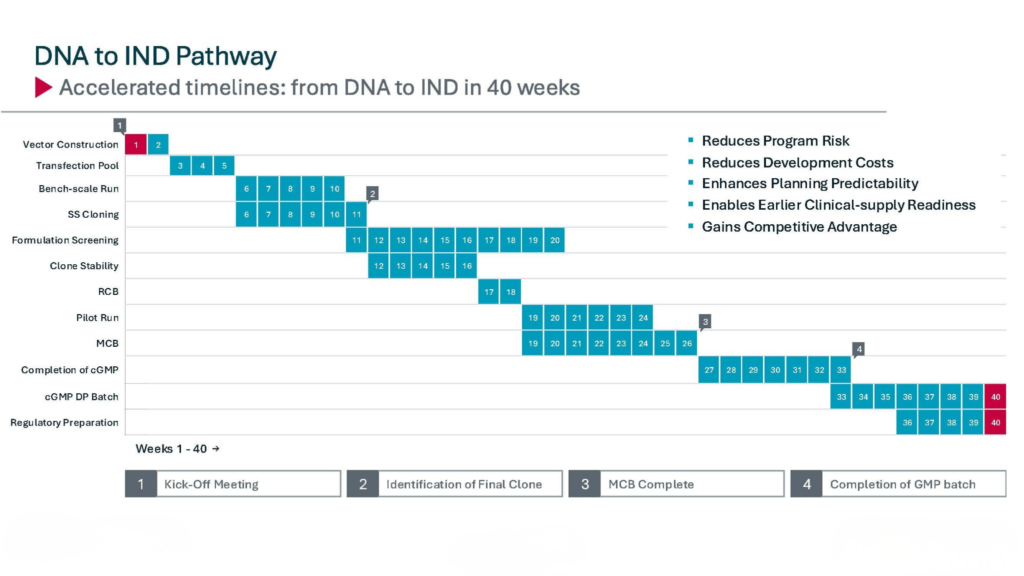
DNA-to-IND timeline at a glance
Unlike traditional development models that rely on sequential handoffs, our timeline reflects a coordinated, platform-based approach where key activities are aligned from the start. The result is not just speed, but predictable execution—reducing the risk of delays during IND-enabling studies and downstream tech transfer.
Start with an early-phase CMC strategy you can defend
Before teams invest heavily in builds and scale-up, it helps to define what “good” looks like for your first-in-human (FIH) path: target product profile, critical quality attributes (CQAs), intended control strategy, and the phase-appropriate level of characterization. This is where timelines are won—or lost—because early CMC decisions determine how efficiently CLD, analytical development, and PD can converge on an IND-ready package. For a deeper dive on how we frame early-phase planning, see our Early Phase White Paper:
CLD: build the foundation for consistent manufacturing
Cell line development (CLD) is more than an upstream milestone—it sets the trajectory for productivity, product quality, and long-term comparability. A fit-for-purpose CLD approach balances speed with selectivity: robust clone screening, genetic stability considerations, and early manufacturability signals that reduce surprises later in development. We’ve captured our perspective in Inside the Science: CLD, a Q&A with Avid subject matter experts:
PD: develop a scalable process with the end in mind
Process development (PD) is where technical intent becomes operational reality. Early PD work should emphasize scalable unit operations, a control strategy that supports manufacturing, and analytics that can meaningfully measure process performance. When PD is aligned to your IND goals, you reduce cycle time during tech transfer and avoid redesigning processes under regulatory pressure. Learn how our teams think about phase-appropriate PD in Inside the Science: PD, a Q&A with an Avid PD SME:
Putting it all together: accelerate without cutting corners
A strong DNA-to-IND program is defined by smart concurrency: CLD informs PD, analytics mature alongside both, and documentation is built as the data arrives—not after the fact. The practical payoff is a cleaner narrative for regulators and a more predictable path to FIH manufacturing. If you’d like to discuss your development strategy, timeline assumptions, or IND CMC expectations, Avid’s teams can help you map the critical path and assemble phase-appropriate data packages to support your IND submission.