Early-Phase Center of Excellence
Costa Mesa, California, USA
Accelerating Early-Stage Biologics
Our dedicated early-phase center of excellence in Costa Mesa, CA is purpose-built to accelerate early-stage biologics from DNA to IND with seamless progression to commercial scale. Designed for speed. Built for scale.

Purpose-built early-stage biologics facility. Integrated development and clinical manufacturing under one roof.
Built Specifically for Early-Stage Biologics
Our Costa Mesa, CA facility is purpose designed to support emerging biopharma programs with the speed, flexibility, and scientific focus required in early development.
Focused on early milestones
Dedicated teams and infrastructure aligned to accelerate cell line development, process development, and clinical manufacturing.
Integrated development and clinical manufacturing
Cell line development, process development and CGMP clinical manufacturing operates within a coordinated platform, reducing handoffs and simplifying execution.
Aligned platforms for easy tech transfer
Development and manufacturing utilize the same core instrumentation and production platforms, supporting smoother tech transfer and reduce scale-up risk.
U.S.-based
Domestic manufacturing capacity aligned with evolving regulatory and supply chain expectations.
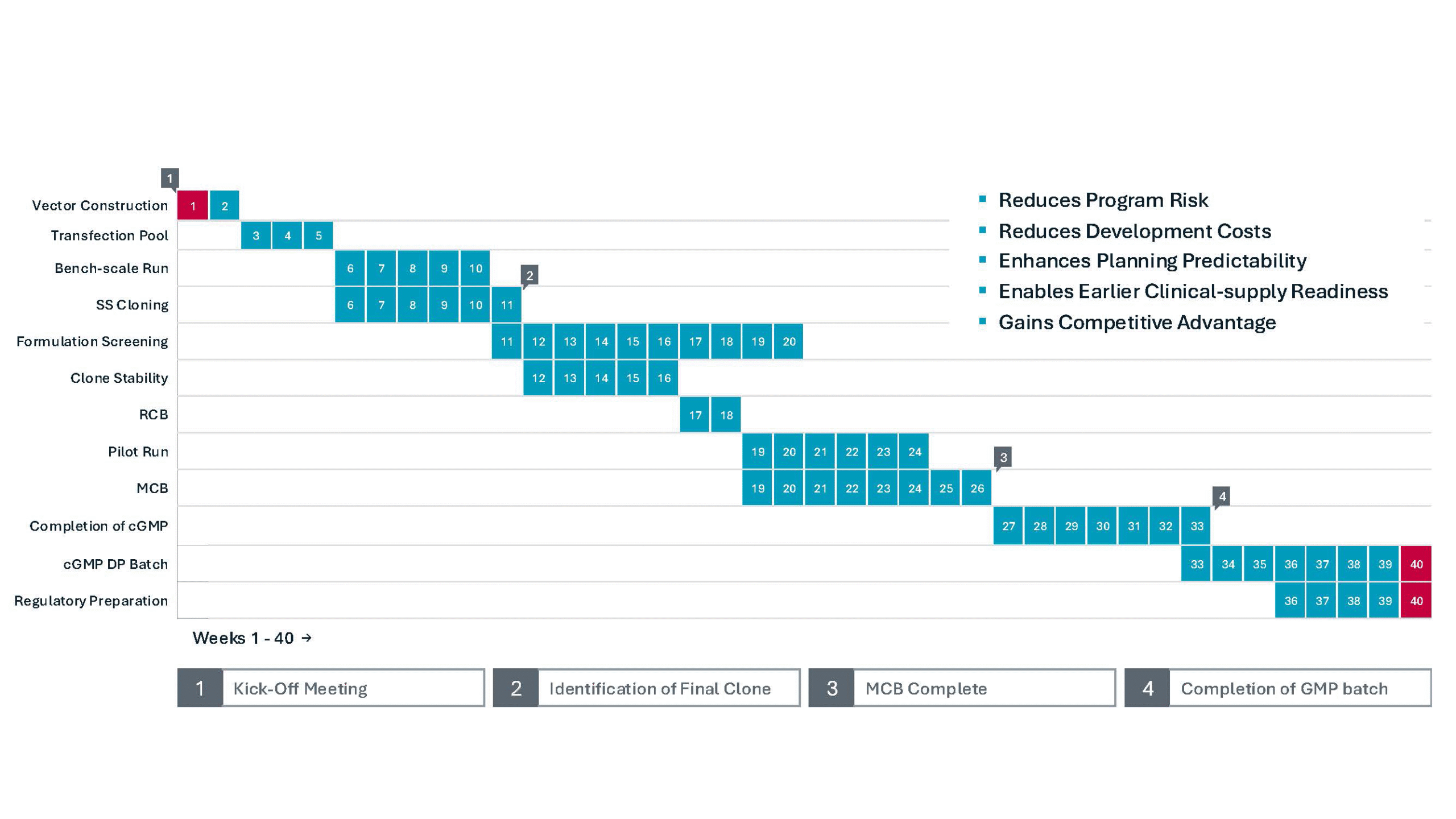
A structured path from DNA to IND
Early-stage programs are built around a disciplined execution and clearly defined milestones. Our integrated development and clinical manufacturing teams support a streamlined progression from cell line development through IND submission approximately 40 weeks*
*Timelines vary based on molecule complexity and program scope.
Early-Stage Capabilities
The Costa Mesa, CA Center of Excellence integrates development and clinical manufacturing capabilities within a single facility to support programs from DNA to IND.

Cell Line Development (CLD)

Process Development (PD)

Analytical and Formulation Development

CGMP Manufacturing

Tech Transfer
Upstream Capacity
Designed for Early-Stage
Our Costa Mesa facility is purpose-built for early-stage biologics development and clinical manufacturing, with scalable mammalian cell culture capacity and modern single-use infrastructure.


Downstream and Analytical Performance
- Single-use downstream platforms
- Integrated development and clinical manufacturing suites
- Dedicated analytical laboratories
- Cleanroom environments aligned with CGMP standards
Capacity & Infrastructure Designed for Early-Stage
Aligned instrumentation and production platforms support predictable tech transfer to commercial manufacturing in our Tustin, CA facility, reducing scale-up complexity and risk.

Quality and Regulatory Readiness
Costa Mesa operates under established CGMP quality systems designed to support IND-enabling activities and early clinical manufacturing with confidence.
- Inspection-ready operations
- IND-enabling Support
- Built for lifecycle continuity
An Early-Stage Team Built Around Your Milestones
Our Early-Stage Center of Excellence is staffed by experienced scientists, engineers, and manufacturing leaders focused specifically on early-stage biologics. Our teams understand the urgency, resource constraints, and milestone pressures facing emerging biologics companies.
Dedicated Program Leadership
Each program is supported by cross functional leaders who remain engaged from cell line development through clinical manufacturing.
Direct Access to Expertise
Clients work directly with the technical and operational decision makers, enabling faster alignment and streamlines execution.
Aligned for Seamless Scale
The early-phase teams collaborate closely with commercial manufacturing counterparts in our Tustin, CA facility to support predictable tech transfer and long-term program success.