Seven Forces Driving Onshoring in Biologics Manufacturing

Policy, trade, and security are prompting proactive moves to the U.S. — before capacity tightens. Biologics manufacturing is being reshaped by a new mix of policy, trade, and security pressures. […]
Five Questions with Rich McAvoy: Scaling Avid with Discipline

We’re excited to welcome Rich McAvoy to Avid Bioservices as our new Chief Business Officer. Rich brings deep industry experience that will help accelerate Avid’s growth while strengthening what we’re […]
Five Questions with Dave Stewart: Scaling Avid for What’s Next

As Avid Bioservices continues to grow and strengthen its foundation for the future, we’re excited to welcome Dave Stewart as our first Chief Technology and Transformation Officer. In this newly […]
Facility-Fit-Driven Process Development for a Monoclonal Antibody (mAb)

Background: A biotech company approached Avid Bioservices to prepare a monoclonal antibody (mAb) process for CGMP manufacturing under a compressed timeline. The client had already developed an initial upstream and […]
Accelerating a Complex Molecule from Cell Line Development to CGMP in 12 Months
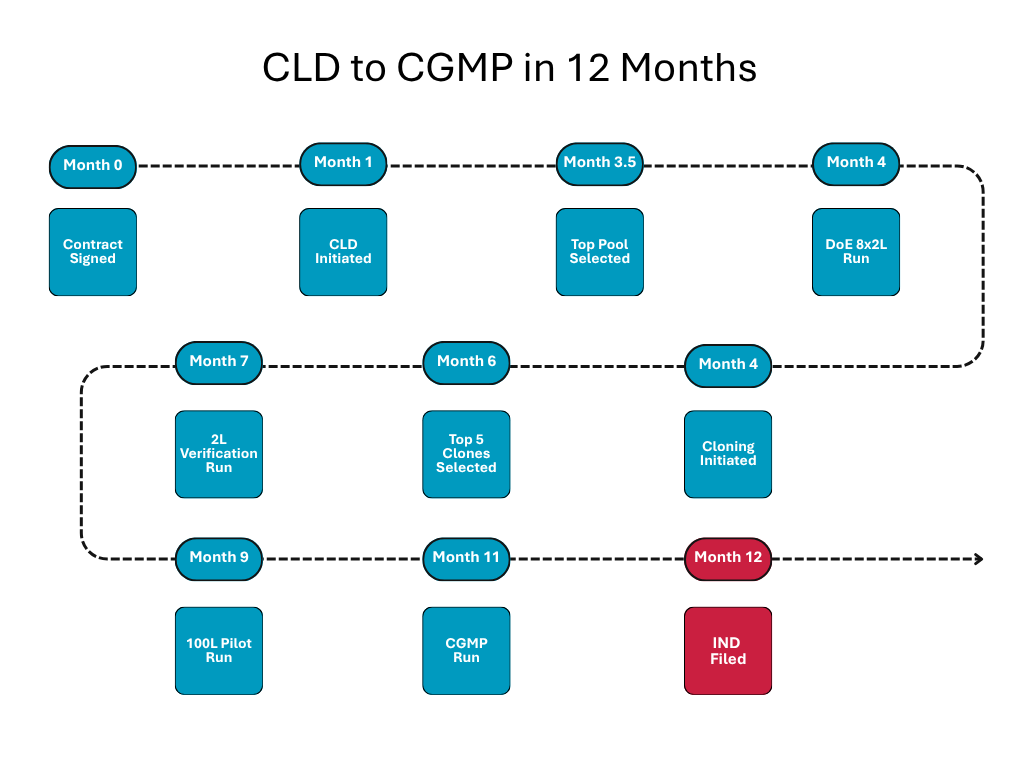
Background: A small, virtual biotech company developing a difficult-to-express bispecific fusion protein needed to advance quickly from discovery to current good manufacturing practice (CGMP) material for clinical studies. The molecule […]
Kenneth Bilenberg on Leading Avid Bioservices and Shaping the Future of Biomanufacturing

By Josh Abbott, Editor, BioProcess Insider Bilenberg spoke to BPI about Avid’s role in biomanufacturing and the outlook for the biopharmaceutical industry. Contract development and manufacturing organization (CDMO) Avid Bioservices recently […]
Shaping the future of biologics manufacturing: Q&A with Avid’s new CEO, Kenneth Bilenberg

Corporate Capabilities Brochure

Process Development Tour

The Power of “What if”
By Nicholas Green, President & CEO We completely transformed the CDMO experience — and the effect is life-changing. Avid Bioservices’ founders have been inspired by the spirit of discovery at […]